Analyzing Gene-to-GP Cosine Similarity
This tutorial demonstrates how to identify and visualize genes that are most relevant to specific gene programs (GPs) in different cell types. This analysis helps answer:
Which genes drive GP activity? - Identify genes with high similarity to a GP’s embedding
How do genes differ between cell types? - Find genes that are differentially associated with a GP across cell types
What is Gene-to-GP Cosine Similarity?
For each gene program, we compute the cosine similarity between:
Each gene’s embedding (learned representation in the GP space)
The GP’s embedding itself
High similarity = Gene is strongly aligned with/represented by that GP Low similarity = Gene is not important for that GP
This reveals which genes are most important for each gene program in eahc cell type
Prerequisites
Completed model training and gene embedding analysis
Gene-to-GP cosine similarities computed and saved in
output_global/gene_embeddings_analysis/
import scanpy as sc
import pandas as pd
from gplearner.Evaluate.downstream import calculate_gene_significance, plot_top_genes
import numpy as np
import anndata as ad
import os
import matplotlib
matplotlib.rcParams['pdf.fonttype'] = 42 # Use TrueType fonts (editable text in PDFs)
2. Load Gene-to-GP Cosine Similarity Data
2.1 Define Loading Function
The gene-to-GP similarity data is stored as AnnData objects where:
Observations (rows): Individual cells
Variables (columns): Genes
Data matrix (X): Cosine similarity between each gene and the GP for each cell
Here, we are examining FOXO3 which was identified as being important in cDC2 in the previous step, but you can replace with any GP of interest.
def load_adata(run):
"""
Load gene-to-GP cosine similarity data for all data splits.
Parameters:
-----------
run : str
Run identifier (e.g., 'run_1', 'run_2', 'run_3') for multi-run averaging
Note: Currently not used in file path, but kept for multi-run compatibility
Returns:
--------
adata : AnnData
Concatenated data with gene-to-GP similarities across all splits
"""
holder = []
base_path = 'path/to/your/folder/07_tutorial_zeng/output_global/gene_embeddings_analysis'
# Load data from train, validation, and test splits
for t in ['train', 'val', 'test']:
# NOTE: This path is specific to GP_FOXO3 analysis
# To analyze other GPs, modify this path (e.g., GP_GATA1_dendritic_cells/)
filename_1 = f'GP_FOXO3_dendritic_cells/gene_to_GP_FOXO3_cosine_similarity_{t}_set.h5ad'
holder.append(
sc.read_h5ad(os.path.join(base_path, filename_1))
)
# Concatenate all splits
adata = ad.concat(holder)
return adata
Multi-run Averaging Option:
In the main Tripso paper, we train the model 3 times with different seeds
Load each run and average the cosine similarities
This reduces stochastic noise from training
Single-run Option (used here):
Faster for exploratory analysis
Suitable for tutorials and initial investigation
# Load data from a single run (tutorial mode)
run1 = load_adata('run_1')
# For publication: load multiple runs and average (commented out)
# run2 = load_adata('run_2')
# run3 = load_adata('run_3')
/software/cellgen/team292/mm58/venvs/lightning2/lib/python3.10/site-packages/anndata/_core/anndata.py:1754: Observation names are not unique. To make them unique, call `.obs_names_make_unique`.
Optionally Average Multiple Runs
If you have multiple training runs, average the similarity scores for increased robustness.
# Option 1: Average across multiple runs (recommended for publication)
# adata = sc.AnnData(
# X = (run1.X + run2.X + run3.X)/3, # Average similarity scores
# obs = run1.obs, # Use metadata from run1 (same across runs)
# var = run1.var # Gene names (same across runs)
# )
# Option 2: Use single run (tutorial mode)
adata = run1
adata
AnnData object with n_obs × n_vars = 16668 × 196
obs: 'length', 'AuthorCellType', 'AuthorCellType_Broad', 'cell_type', 'Sorting', 'Study', 'donor', 'sex', 'development_stage', 'age_group', 'n_counts', 'idx', 'batch_key', 'cell_type_id', 'age_group_id', 'batch_key_id'
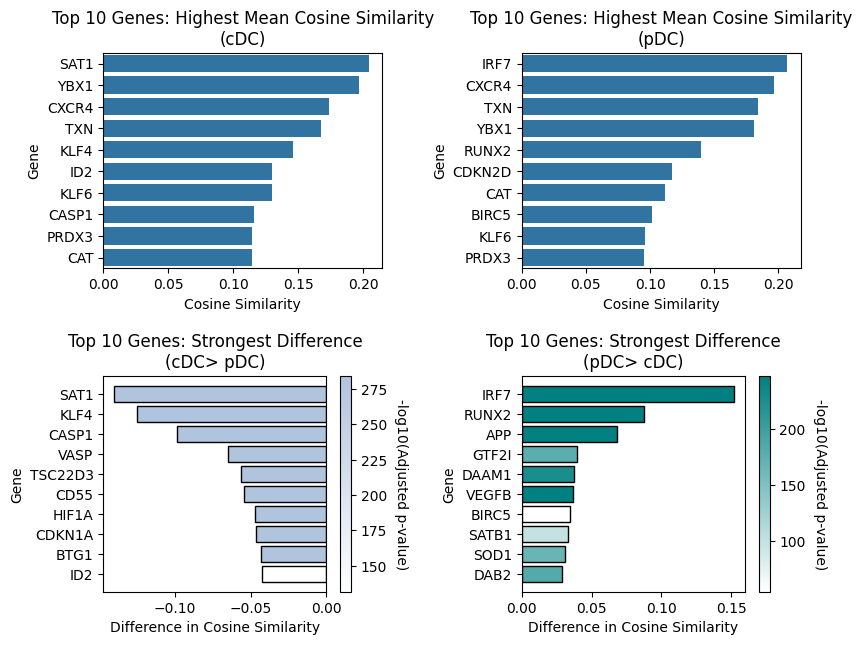
3. Comparison: pDC vs. cDC
Goal: Identify genes that are differentially similar to GP_FOXO3 between plasmacytoid DCs and conventional DC.
Parameters:
obs_value_ref="cDC": Reference population (baseline)obs_value_query="pDC": Query population (comparison)adata_gene_threshold=0.9: drop genes which are 0 in >90% of cells
Interpretation:
Positive values = Genes with higher GP_FOXO3 similarity in pDC
Negative values = Genes with higher GP_FOXO3 similarity in cDC2
# Calculate differential gene-to-GP similarity between pDC and cDC2
res = calculate_gene_significance(
adata,
obs_col="AuthorCellType_Broad", # Column containing cell type annotations
obs_value_ref="cDC", # Reference cell type (baseline)
obs_value_query="pDC", # Query cell type (comparison)
adata_gene_threshold=0.9, # Filter: keep genes detected in ≥90% of cells
)
Visualize Top Differentially Similar Genes (pDC vs. cDC2)
Create a bar plot showing the top genes with differential GP_FOXO3 similarity between pDC and cDC2.
Visualization Parameters:
topn=10: Show top 10 genesshow_significance=False: Don’t add stars for genes reaching statistically significancecolor_scheme='significance': Color by significance (-log10 adjusted p-value)significance_palette: Custom colors for significance (darker = more significant)palette_as_gradient=True: Use significance color gradient rather than color blocks
# Visualize top genes differentially associated with GP_FOXO3 in pDC vs. cDC2
plot_top_genes(
res,
obs_value_ref="cDC",
obs_value_query="pDC",
show_significance=False, # Hide significance stars
color_scheme='significance', # Color by effect size
significance_palette=['lightsteelblue', 'teal'], # Color gradient
palette_as_gradient=True, # Smooth gradient
topn=10, # Display top 10 genes
figsize=(9, 7),
# save_to='GP_FOXO3_pDC_vs_cDC2.pdf' # Uncomment to save figure
)
/nfs/team361/mm58/gplearner/gplearner/Evaluate/downstream.py:2196: UserWarning: set_ticklabels() should only be used with a fixed number of ticks, i.e. after set_ticks() or using a FixedLocator.
axs[1, 0].set_yticklabels(yticklabels)
/nfs/team361/mm58/gplearner/gplearner/Evaluate/downstream.py:2281: UserWarning: set_ticklabels() should only be used with a fixed number of ticks, i.e. after set_ticks() or using a FixedLocator.
axs[1, 1].set_yticklabels(yticklabels)